Smartview™ Remote Monitoring
Makes it easy. Makes it smarter.
SMARTVIEW remote monitoring system takes you to the next level in patient management thanks to its simplicity of use and comprehensive automatic reporting abilities.
Features
The SMARTVIEW remote monitoring system allows monitoring of MicroPort Cardiac electronic implantable devices (CIEDs). It supports monitoring of devices with wireless communication as well as those with inductive telemetry.
The system is designed to help healthcare professionals with managing the high workload associated with cardiac device follow-ups.
SMARTVIEW simplifies patient care and ensures the timely delivery of relevant device information. The SMARTVIEW remote monitoring system implements a multi-tiered security system that safeguards patient personal data according to applicable regulations.
This product may not be for sale in your country or your area. For further information on product availability, please contact your local representative. Take caution: Federal law (USA) restricts this device to sale by or on the order of a physician. For indications, contraindications, precautions and warnings for each device, please refer to the product manual.

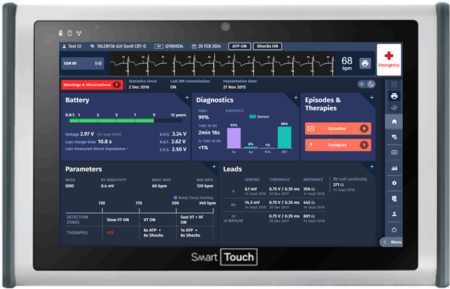
SmartTouch™ Programmer
A modular system based on a medical-grade tablet
The SmartTouch is the embodiment of the state-of-the-art for programmers.
Features
- Light weight for mobile usage
- Modular design facilitates versatile adaptation in different clinical environments
- Bluetooth connectivity allows implantable devices and accessories to wirelessly communicate with the programmer
Not available for distribution or sale in the USA

SMARTTOUCH XT™ PROGRAMMER
Designed with you, for you
The SmartTouch XT™ is a programmer1 designed with clinicians for clinicians, which reaches an unprecedented level in ergonomics and ease of use.
Features
- User-driven design ensures excellent response to actual customers’ needs
- Smart positioning of the telemetry head helps the performance of inductive communication
- Light weight for mobile usage
- Modularity facilitates versatile adaptation to different clinical environments
- Bluetooth connectivity allows implantable devices and accessories to wirelessly communicate with the programmer
Caution: Federal law (USA) restricts this device to sale by or on the order of a physician. Refer to user’s manual for intended use and relevant warnings, precautions, side effects, and contraindications.
1. SmartTouch XT™ is the commercial name of the system composed by SmartTouch™ tablet, CPR4 inductive head, SmartTouch XT™ docking station and additional optional accessories.

Orchestra™ Plus Programmer
A programmer designed by users
The Orchestra Plus Programmer was designed in close collaboration with cardiologists in order to provide a comprehensive system that serves the needs of a clinical practice environment.
Features
The Orchestra Plus programmer is an all-in-one classic programmer used to program MicroPort® CRM pacemakers and defibrillators. It supports the full range of implanted devices including those of the former ELA/Sorin Group.
Caution: Federal law (USA) restricts this device to sale by or on the order of a physician.

Tachy User Interface
Discover the Tachy User interface
Features
- Modern design: Our Tachy UI boasts a modern and visually appealing design
- Easy and efficient navigation: An intuitive and efficient navigation structure, ensuring users can easily find what they are looking for without unnecessary clicks
- Personalization option: Bright and dark modes display, allowing users to tailor the interface to their specific needs and preferences
- Adapted to SmartTouch XT™ programmer: A fully responsive UI, that provides a consistent and optimal experience when using our programmer